| Synthesis of High Performance Polymer Materials for Field Effect-Transistors |
| From: PublishDate:2012-10-11 Hits: |
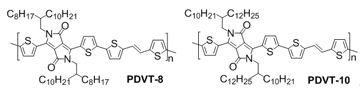
Huajie Chen, Yunlong Guo, Yan Zhao, Gui Yu* and Yunqi Liu* yugui@iccas.ac.cn, liuyq@iccas.ac.cn Beijing National Laboratory for Molecular Sciences, Institute of Chemistry, Chinese Academy of Sciences, Beijing 100190, P. R. China Solution-processed organic semiconductors to replace amorphous silicon as the active layer in field-effect transistors (FETs) have attracted great attention due to their potential applications in flexible and low-cost E-paper, smart card, radio frequency identification, and displays.Recently, significant progress has been made in the performance of solution-processed FETs based on both single-crystalline (5~30 cm2 V−1s−1) and thin-film (2~3 cm2 V−1s−1) of organic small-molecule semiconductors. However, the thin-film uniformity of the small molecules usually varies from area to area due to their high crystalline properties, thus limiting their large-scale applications. In contrast, soluble polymer semiconductors meet well with the requirements of fast solution process for organic optoelectronic applications because of their great advantages in terms of excellent mechanical flexibility and solution processability relative to the soluble small-molecules and single crystals. However, the mobility in polymeric FETs is generally low due to the poor packing of those materials. Recently, researchers in Key Laboratory of Organic Solids, Institute of Chemistry, Chinese Academy of Sciences have made great progress in this field. The develop a series of highly π-extended copolymers containing diketopyrrolopyrrole (DPP) and (E)-2-(2-(thiophen-2-yl) vinyl)thiophene (TVT) moieties (Figure 1). The as-synthesized copolymer PDVT-8 with short 2-octyldodecyl side-chains showed a high field-effect mobility of 2.0~4.5 cm2 V−1s−1, with a current on/off ratio of 105~107. Meanwhile, the copolymer PDVT-10 with long alkyl side-chains (2-decyltetradecyl groups) exhibited an unprecedented high mobility of up to 8.2 cm2 V−1s−1, a current on/off ratio of 105~107, and an outstanding environmental stability under 20% air humidity (Figure 2). The mobility decreased about 33% in the first 7 days and negligible changes were observed in the subsequent 100 days.
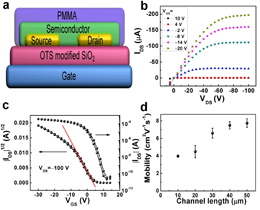
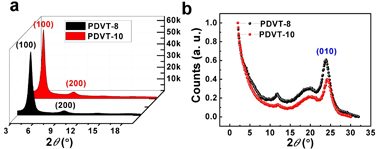
Figure 1. Chemical structures and synthetic routes of the polymers Figure 2. (a) Devices structure of the polymer FETs with a PMMA encapsulation layer. (b) Output and (c) Transfer characteristics of the PDVT-10 based FET device with W = 1400 μm and L = 50 μm, exhibiting a hole mobility of 8.0 cm2 V−1s−1 at VDS= –100 V. (d) The mobility distributions based on different channel lengths. To understand the relationships between the film morphology/crystallinity and device performance, the polymer thin films were characterized by GIXRD in Beijing Synchrotron Radiation Facility (BSRF). The out-of-plane GIXRD patterns (Figure 3a) have demonstrated that both PDVT-8 and PDVT-10 thin films exhibit a lamellar stacking structure, with interlayer distances of 19.44 Å and 21.11 Å, respectively. Because these (h00) peaks are not seen in the in-plane GIXRD patterns (Figure 3b), it is concluded that the polymers were oriented perpendicular to the dielectric layer. This orientation is well-known to achieve high carrier mobility because of the intermolecular interactions parallel to the direction of the channel current flow. Both polymer films exhibit very closed stacking with the π-π stacking distances of 3.72 Å for PDVT-8 and 3.66 Å for PDVT-10, respectively. Such small π-π distances between polymer backbones suggests that the strongly intermolecular interactions exist, which can be accounted for the π-π stacking of highly π-extended polymer backbones and the strong D–A interactions.
Figure 3. (a) Grazing incidence X-ray scattering pattern (out-of-plane) of polymer PDVT-8 and PDVT-10 films annealed at 180 °C. (b) In-plane X-ray scattering pattern of polymer PDVT-8 and PDVT-10 films annealed at 180 °C. The result was published in Advanced Materials (2012, 24, 4618–4622)and was selected as cover.
Acknowledgements: The GIXRD data were obtained at 1W1A, Beijing Synchrotron Radiation Facility. The authors gratefully acknowledge the assistance of scientists of Diffuse X-ray Scattering Station during the experiments. This work was supported by the National Natural Science Foundation of China (20825208, 60911130231, 61101051 and 21021091), and the Major State Basic Research Development Program (2011CB808403, 2011CB932303, 2009CB623603), and Chinese Academy of Sciences. |
|
|
| Chinese
- Synthesis of High Performance Polymer Materials for Field Effect-Transistors
- Surfactant molecular aggregates in green solvents
- GIXRD has played an important role in the characterization of organic thin-film transistors
News and Events
Home /
Copyright © 2011 - 2012 Beijing Synchrotron Radiation Facility