| Silver Ion-Mediated Heterometallic Three-Fold Interpenetrating Uranyl–Organic Framework |
| From: PublishDate:2016-06-03 Hits: |
Uranyl coordination chemistry as well as its speciation is of great significance in the nuclear industry in terms of nuclear waste separation and disposal, meanwhile the intensive study on it will help to explore novel uranyl-organic frameworks with intriguing structure or function, i.e. uranyl-organic interpenetrating networks. However, the design and synthesis of uranyl-organic interpene+trating networks bearing heterometal is still a tough objective to be achieved. A team from Multi-discipline Research Center, Institute of High Energy Physics has successfully prepared the first silver ion-mediated heterometallic three-fold interpenetrating uranyl-organic framework. Their research has been published on November 16th, 2015 in Inorganic Chemistry.
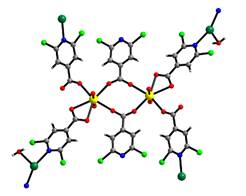
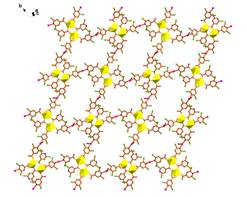
Heterometallic interpenetrating uranyl-organic framework obtained based on Beijing Synchrotron Radiation Facility (BSRF). In this compound, uranyl dimer with six carboxylate ligands acted as building unit and further was linked by Ag+ ions from four directions, finally affording a heterometallic interpenetrating network. This intriguing structure was formed mainly through the coordination bonding of carboxylate and pyridine groups, while the structural halogenation also plays an important role in the stabilization of it. The team has found that the stable symmetrically halogenated isonicotinic acid could bind to the uranyl center, forming an isolated carboxylate-bridged uranyl dimers halogen-bonded with each other in a work they reported previously. Their further research demonstrated that the addition of Ag+ ion into this uranyl-isonicotinate system can alter effectively uranyl coordination pattern, leading to the establishment of a novel heterometallic interpenetrating uranyl-organic framework. Nevertheless, other metal ions, i.e. Cu2+, Zn2+, Co2+, Ni2+, didn’t work like Ag+ ion. Crystallographic analysis showed that a (4, 4)-connected uranyl dimer building unit with six carboxylate ligands was formed by the induction of Ag+ ion, which was further linked with Ag+ ions from four directions and finally afforded a heterometallic three-fold interpenetrating network. Moreover, the mediation of Ag+ ion always works whatever before or after the coordination of uranyl by halogenated isonicotinate. The research provides a different design route for exploring the establishment of heterometallic interpenetrating uranyl-organic framework, and synchrotron sources have helped the team to overcome the technical problem of determing the molecular structure of actinide complexes in the size of several microns. "Uranyl complexes exhibit a variety of structures in different reaction systems, and it is still difficult to control or predict concisely the formation of expected species. Therefore, the research on actinide complexes is on the way, focusing on the characterization of molecular structures and functions of many newly-prepared uranyl complexes so as to dig out the formation rules of them and then achieve a controllable synthesis. More brilliant synchrotron beams would surely help to examine those smaller actinide-based crystals." explains Wei-qun Shi, the team leader and the professor of Multi-discipline Research Center, Institute of High Energy Physics. Article: Lei Mei, Qun-yan Wu, Shu-wen An, Zeng-qiang Gao, Zhi-fang Chai and Wei-qun Shi* Silver Ion-Mediated Heterometallic Three-Fold Interpenetrating Uranyl–Organic Framework. Inorg. Chem., 2015, 54 (22), 10934–10945. |
|
|
| Chinese
- Beamline 1W1 of BSRF started to runoperate in the couplingparasitic mode of BEPCII
- Synthesis of High Performance Polymer Materials for Field Effect-Transistors
- Surfactant molecular aggregates in green solvents
- GIXRD has played an important role in the characterization of organic thin-film transistors
Copyright © 2011 - 2012 Beijing Synchrotron Radiation Facility